DFG founded research groups » Research Group Lippmann » Research
Function-to-structure analysis of synapses to understand presynaptic vesicular dynamics and plasticity
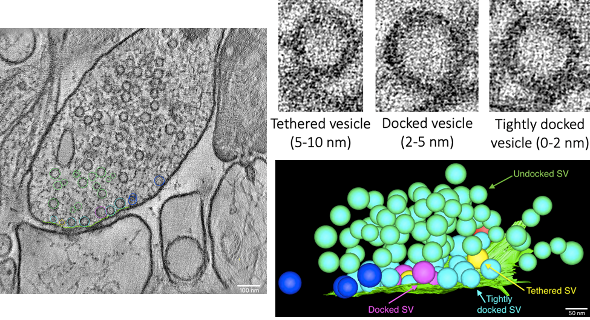
Synaptic transmission is fundamental for nerve cell communication, brain activity and cognition. Brain activity and cognition are not static but dynamic processes subserving learning and environmental adaptation. Until recently, it was possible to study synaptic transmission only either functionally by using electrophysiology, with which one can measure synaptic currents in nerve cells, or by imaging snapshots of synaptic structures using high-resolution electron microscopy. By using a technique called zap-and-freeze, biological tissue like acute brain slices can be electrically stimulated and subsequently high-pressure frozen within milliseconds. Highly dynamic processes, such as presynaptic vesicle fusion and endocytosis can thus be resolved spatiotemporally with electron microscopy. Using this approach in acute brain slices, we investigate vesicular dynamics, such as occurring during presynaptic plasticity, in space and time. Electron tomography further enables us to three-dimensionally reconstruct larger parts of synapses for in-depth analyses of vesicular docking states and fusion pits.
Cellular mechanisms underlying network oscillations in epilepsy-related diseases
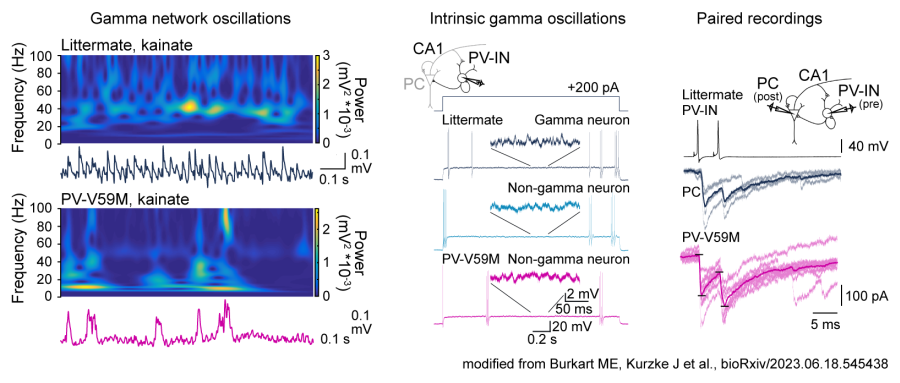
Hippocampal network oscillations, such as gamma oscillations and sharp wave ripples, are fundamental for information perception, processing and memory consolidation in the brain. In epilepsy-related diseases, network oscillations are often disrupted and could thus contribute to cognitive dysfunctions of patients. We aim to better understand the cellular mechanisms underlying impaired hippocampal network oscillations in acquired and congenital epilepsy-related diseases. For this, we use patch-clamp electrophysiology for studying synaptic and intrinsic cellular activities as well as local field potential recordings to characterize changes in network oscillations in acute brain slices and in vivo. We are particularly interested in functional changes of parvalbumin-positive fast-spiking interneurons (PV-IN), inhibitory neurons that are key players for generating gamma oscillations and sharp wave ripples as well as for promoting epileptic activity in case of their dysfunction.

