Department III Hirrlinger » Research
The brain is the most complex organ of mammals which allows both control of body functions as well as execution of complex behaviors. It consists of two major groups of cells, neurons and glial cells; and proper brain function requires the close and tightly controlled cooperation of all these cells.
Three major types of glial cells exist in the central nervous system: astrocytes, oligodendrocytes and microglial cells. Astrocytes contribute to many functions of the brain including metabolism, ion homeostasis, blood flow regulation and neurotransmitter clearance. They are active partners of neurons in information processing participating in regulation of synaptic function and plasticity. Oligodendrocytes form the myelin sheats around axons, thereby allowing rapid saltatory action potential propagation. However, recent research indicates that oligodendrocytes play a much more active role in interaction with axons and other cells than previously appreciated. Finally, microglial cells are the immune cells of the central nervous system contributing to tissue homeostasis and repair.
Research in our department focusses on several aspects including glial cell physiology, the communication and interaction of glial cells and neurons as well as the development of new technologies:
1) Brain energy metabolism
The function of the brain crucially depends on an appropriate supply of energy. Astrocytes have long been implicated in the supply of energy and blood flow regulation; however, many questions regarding mechanisms and regulation remained open so far. Especially, we are interested in how cellular signaling events regulate metabolism and how metabolic factors feedback to modulating signaling processes. We address these issues using a combination of modern imaging technologies including 2-photon imaging, fluorescent sensors for metabolites and calcium imaging. Recently, we could e.g. show that dopamine signaling regulates the NAD+/NADH redox state in astrocytes which in turn modifies Ca2+ signals (Requardt et al., 2010; 2012).
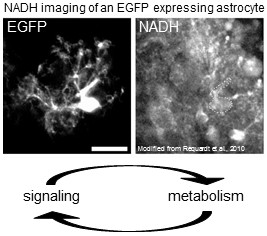
2) Astrocyte processes: dynamics and regulation
A prerequisite for functional interactions of astrocytes and neurons is a close morphological interaction. Astrocytic processes are indeed in close contact to blood vessels, other brain cells and synapses. However, these processes are not static structures but are formed and retracted within minutes (Hirrlinger et al., 2004). We aim at understanding the mechanisms and regulation of this interaction, e.g. by time-lapse imaging of astrocytic dynamics as well as by selectively deleting cell-adhesion associated proteins in vivo (Winkler et al., 2013)
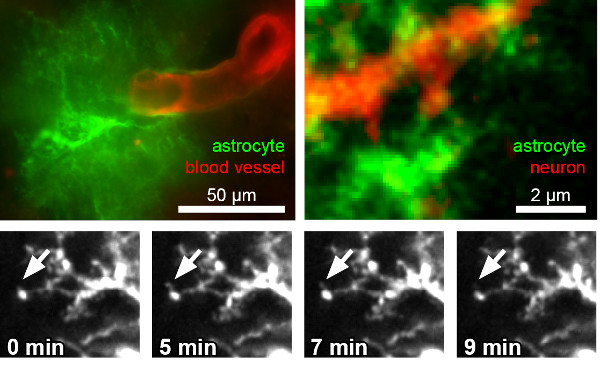
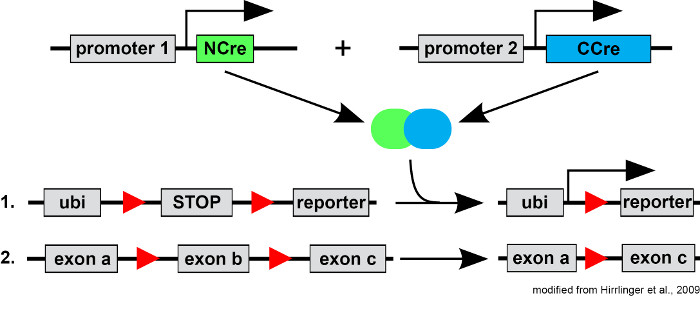
3) Precise targeting of cell-populations using split-Cre
Conditional and cell-type specific gene targeting using the Cre-LoxP-technology has provided major advances in our knowledge of cell fate and cell-type specific gene function. However, the precision of cell targeting crucially relies on the specificity of the promoter used to drive Cre expression. To refine the resolution of Cre mediated cell targeting, we have developed the split-Cre-system which allows genetically defining cell populations by the simultaneous activity of two promoters (Hirrlinger et al., 2009). We showed that a subpopulation of astrocytes is derived from precursor cells expressing both the glial fibrillary acidic protein (GFAP)- as well as the proteolipid (PLP) protein- promoter (Hirrlinger et al., 2009). Furthermore, split-Cre allowed defining adult neural stem cells in vivo (Beckervordersandforth et al., 2010). Current projects aim at targeting highly specifically selected populations of glial cells and neurons to address the fate and function of these cells.
Funding
Research in in our group is funded by the German Research Council (Deutsche Forschungsgemeinschaft, DFG) as well as the German Diabetes Society (DDG).
Selected publications
(full list of publications) http://www.ncbi.nlm.nih.gov/pubmed/?term=hirrlinger-j
-
Köhler S, Winkler U, Hirrlinger J. (2021)Heterogeneity of Astrocytes in Grey and White Matter.Neurochem Res. 46(1):3-14.
-
Trevisiol A, Kusch K, Steyer AM, Gregor I, Nardis C, Winkler U, Möbius W, Werner HB, Nave KA, Hirrlinger J (2020)Structural myelin defects are associated with low axonal ATP levels but rapid recovery from energy deprivation in a mouse model of spastic paraplegia.PLoS Biol. 18: e3000943.
-
Köhler S, Schmidt H, Fülle P, Hirrlinger J, Winkler U (2020)A dual nanosensor approach to determine the cytosolic concentration of ATP in astrocytes.Front. Cell. Neurosci. 14: 565921.
-
Hirrlinger J, Marx G, Besser S, Sicker M, Köhler S, Hirrlinger PG, Wojcik SM, Eulenburg V, Winkler U, Hülsmann S (2019)GABA-Glycine Cotransmitting Neurons in the Ventrolateral Medulla: Development and Functional Relevance for Breathing.Front. Cell. Neurosci. 13: 517.
-
Baeza-Lehnert F, Saab AS, Gutiérrez R, Larenas V, Díaz E, Horn M, Vargas M, Hösli L, Stobart J, Hirrlinger J, Weber B, Barros LF (2019)Non-Canonical Control of Neuronal Energy Status by the Na+ Pump.Cell Metab. 29: 668-680.e4.
-
Köhler S, Winkler U, Sicker M, Hirrlinger J (2018)NBCe1 mediates the regulation of the NADH/NAD+ redox state in cortical astrocytes by neuronal signals.Glia 66: 2233-2245.
-
Schleinitz D, Seidel A, Stassart R, Klammt J, Hirrlinger PG, Winkler U, Köhler S, Heiker JT, Schönauer R, Bialek J, Krohn K, Hoffmann K, Kovacs P, Hirrlinger J. (2018)Novel Mutations in the Asparagine Synthetase Gene (ASNS) Associated With Microcephaly.Front. Genet. 9: 245
-
Trevisiol A, Saab AS, Winkler U, Marx G, Imamura H, Möbius W, Kusch K, Nave KA, Hirrlinger J (2017)Monitoring ATP dynamics in electrically active white matter tracts.Elife 6: e24241.
-
Saab AS, Tzvetavona ID, Trevisiol A, Baltan S, Dibaj P, Kusch K, Mobius W, Goetze B, Jahn HM, Huang W, Steffens H, Schomburg ED, Perez-Samartin A, Perez-Cerda F, Bakhtiari D, Matute C, Lowel S, Griesinger C, Hirrlinger J, Kirchhoff F, Nave KA (2016)Oligodendroglial NMDA Receptors Regulate Glucose Import and Axonal Energy Metabolism.Neuron 91: 119-132
-
Winkler U, Seim P, Enzbrenner Y, Köhler S, Sicker M, Hirrlinger J (2017)Activity dependent modulation of intracellular ATP in cultured cortical astrocytes.J. Neurosci. Res. 95: 2172-2181.
-
Winkler U, Hirrlinger J. (2015)Crosstalk of Signaling and Metabolism Mediated by the NAD+/NADH Redox State in Brain Cells.Neurochem Res. 40(12): 2394-2401
-
Hirrlinger J, Nave KA.(2014)Adapting brain metabolism to myelination and long-range signal transduction.Glia. 2014; 62(11):1749-61
-
Winkler U, Hirrlinger PG, Sestu M, Wilhelm F, Besser S, Zemljic-Harpf AE, Ross RS, Bornschein G, Krügel U, Ziegler WH, Hirrlinger J (2013)Deletion of the cell adhesion adaptor protein vinculin disturbs the localization of GFAP in Bergmann glial cells.Glia 61: 1067-1083
-
Requardt RP, Hirrlinger PG, Wilhelm F, Winkler U, Besser S, Hirrlinger J (2012)Ca2+ signals of astrocytes are modulated by the NAD⁺/NADH redox state.J. Neurochem. 120: 1014-1025
-
Beckervordersandforth R, Tripathi P, Ninkovic J, Bayam E, Lepier A, Stempfhuber B, Kirchhoff F, Hirrlinger J, Haslinger A, Lie DC, Beckers J, Yoder B, Irmler M, Götz M (2010)In vivo fate mapping and expression analysis reveals molecular hallmarks of prospectively isolated adult neural stem cells.Cell Stem Cell 7:744-758
-
Hirrlinger J, Scheller A, Hirrlinger PG, Kellert B, Tang W, Wehr MC, Goebbels S, Reichenbach A, Sprengel R, Rossner MJ, Kirchhoff F (2009)Split-Cre Complementation Indicates Coincident Activity of Different Genes In vivo.PLOS One 4: e4286
-
Hirrlinger J, Hülsmann S, Kirchhoff F (2004)Astroglial processes show spontaneous motility at active synaptic terminals in situ.Eur. J. Neurosci. 20: 2235-2239 Comment in “Research Highlights” (2004) Nat. Rev. Neurosci. 5: 828-829
Books
-
Hirrlinger J, Waagepetersen HS (eds.) (2014)Brain Energy MetabolismNeuromethods, vol. 90 (2014) Springer Science+Business Media New York

