Department I Hallermann » Techniques
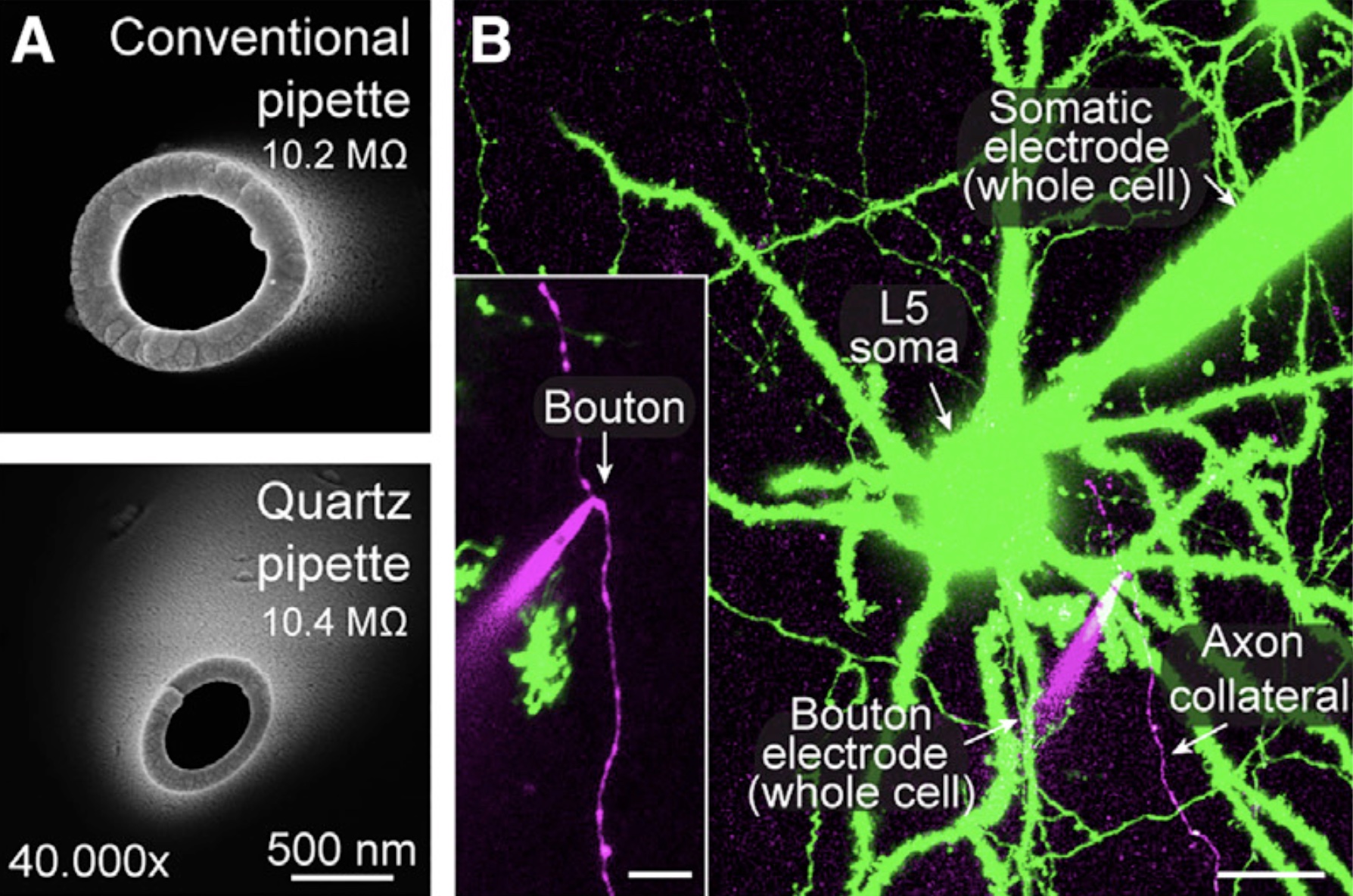
Presynaptic patch-clamp recordings from small CNS boutonsTo investigate the presynaptic mechanisms of chemical synaptic transmission, we perform patch-clamp recordings from presynaptic nerve terminals in acute brain slices, including typical small boutons in the neocortex (Ritzau-Jost et al., Cell Reports, 2021), dopaminergic axons in the striatum (Liu et al., Science, 2022), mossy fiber boutons in the cerebellum (Ritzau-Jost et al., Neuron, 2014), and mossy fiber boutons in the hippocampus (Hallermann et al., PNAS, 2003). The figure shows electron microscopic images of the tip of borosilicate and quartz glass pipettes (A) and a two-photon image of a paired whole-cell recording from the soma and bouton of a layer 5 pyramidal neuron (B; scale bar 20 µm and 5 µm for the inset; Ritzau-Jost et al., Cell Reports, 2021). |
 |
Paired pre- and postsynaptic recordingsWe established paired recordings from cerebellar mossy fiber boutons and postsynaptic granule cells to investigate synaptic transmission at sub-millisecond resolution. This allows combining presynaptic capacitance measurements and deconvolution methods. The figure shows a paired pre- and postsynaptic recording (top; magenta and green, respectively; scale bar 10 µm) and the superposition of membrane capacitance and deconvolution-based cumulative vesicle fusion rate (bottum; Ritzau-Jost et al., Neuron, 2014). |
  |
High-resolution quantitative two-photon calcium imagingQuantitative two-photon calcium imaging with various calcium indicators were established to better understand presynaptic high-frequency vesicular release (figure showing presynaptic calcium concentration during 300-Hz-transmission; Delvendahl et al., PNAS, 2015). |
 |
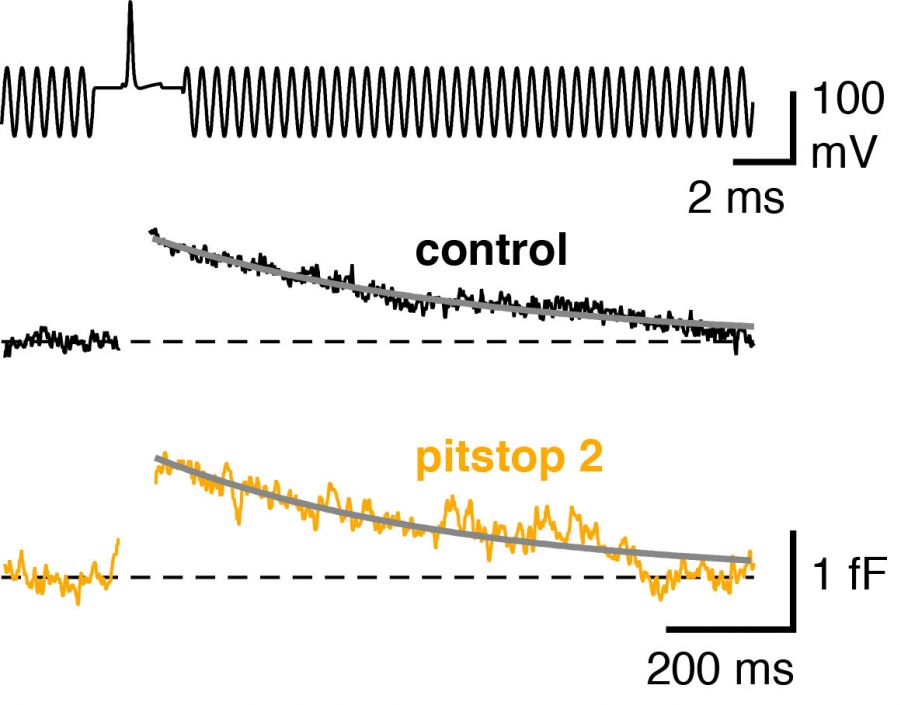
Low-noise capacitance measurements with quartz-glass pipettesWe perform presynaptic capacitance measurements with quartz-glass pipettes to resolve exo- and endocytosis evoked by single action potentials in CNS synapses (Figure showing the capacitance transient evoked by single action potentials after pharmacological blockade of clathrin mediated endocytic pathway; Delvendahl et al., Neuron, 2016). |
 |
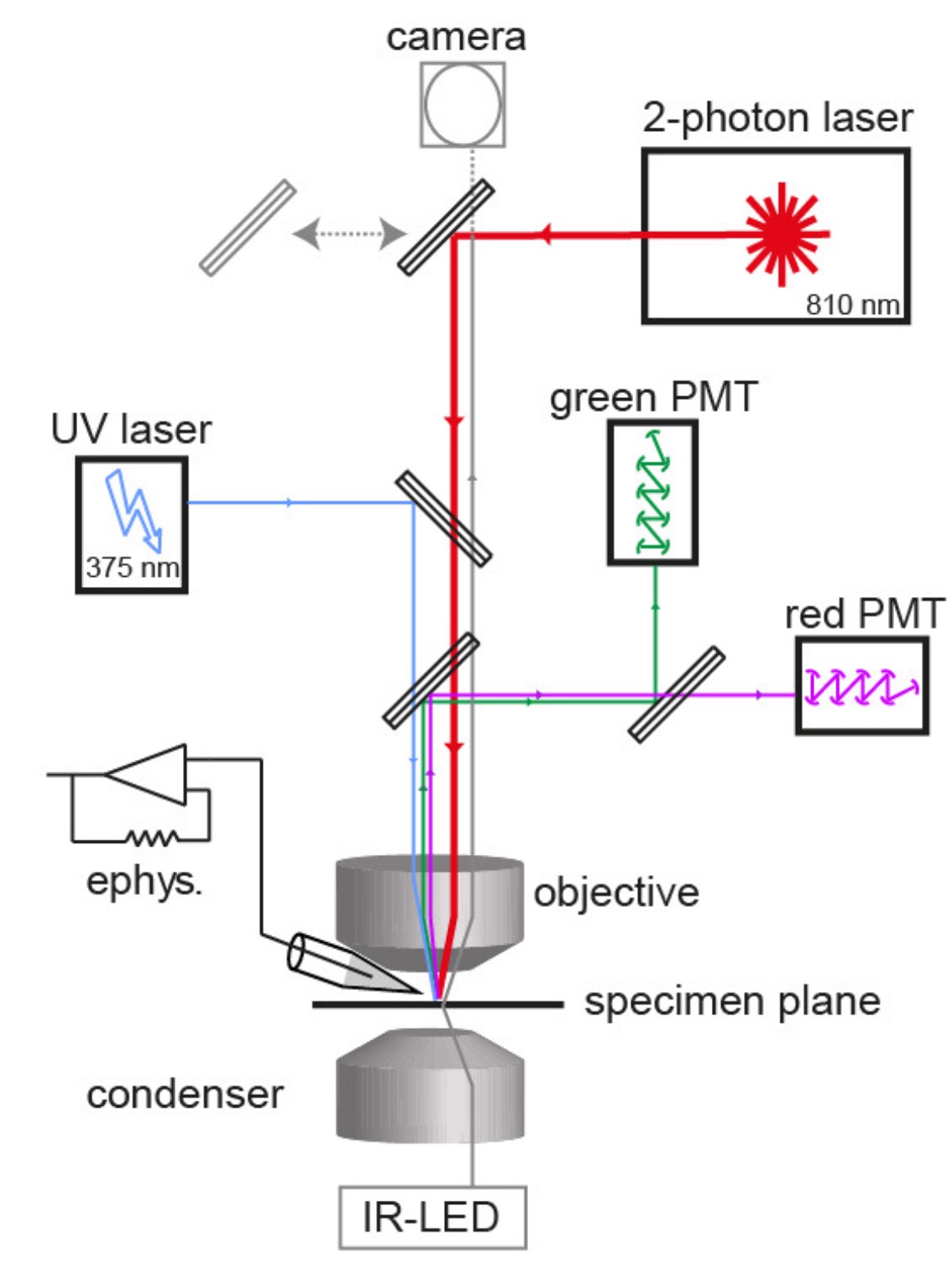
Calcium uncagingWe combine quantitative two-photon calcium imaging with wide-field UV excitation to measure the calcium sensitivity of vesicle fusion. The figure illustrates the experimental setup with the light path of the two-photon laser illumination (red line), the UV laser illumination (blue line), the electrophysiology amplifier (‘ephys.’), the red and green gate-able photomultiplier tubes (PMTs), and infrared LED illumination (gray line; Eshra et al., eLife, 2021). |
 |
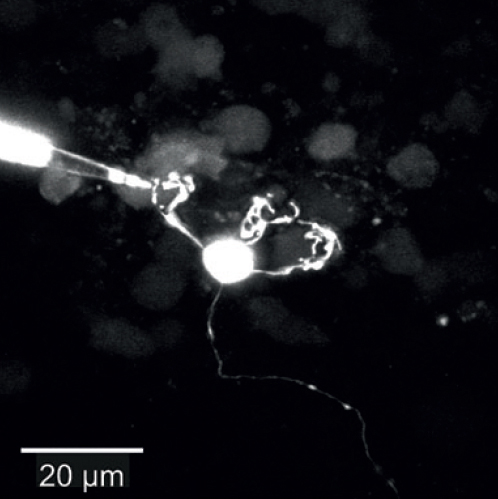
Dendritic patch-clamp recordingsTo understand dendritic integration, we use patch-clamp recordings from dendrites of cerebellar granule cells (see Figure; Delvendahl et al., Front Cell Neurosci, 2015) or cortical layer 5 pyramidal neurons (Hallermann et al., Nat Neurosci, 2012). |
 |
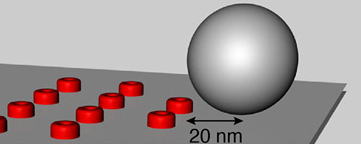
Modeling of synpases and cellsWe analyze calcium diffusion, calcium buffering, and vesicle fusion using the CalC simulation environment (web.njit.edu/~matveev; Figure illustrating a synaptic vesicle in grey and calcium channels in red; Delvendahl et al., PNAS, 2015). Channel gating and cellular excitability are simulated with Neuron (www.neuron.yale.edu; Hallermann et al., Nat Neurosci, 2012). Quantal short term plasticity is implemented in C++ (Hallermann et al., Neuron, 2010; Hallermann et al., J Neurosci, 2010). |
 |
Spiking neural networksTo investigate computation on the network level we use the neural networks of integrate and fire neurons. The figure illustrates the neuronal network model of the cerebellar cortex while learning a temporal sequence of spiking (Straub et al., eLife, 2020). |
 |
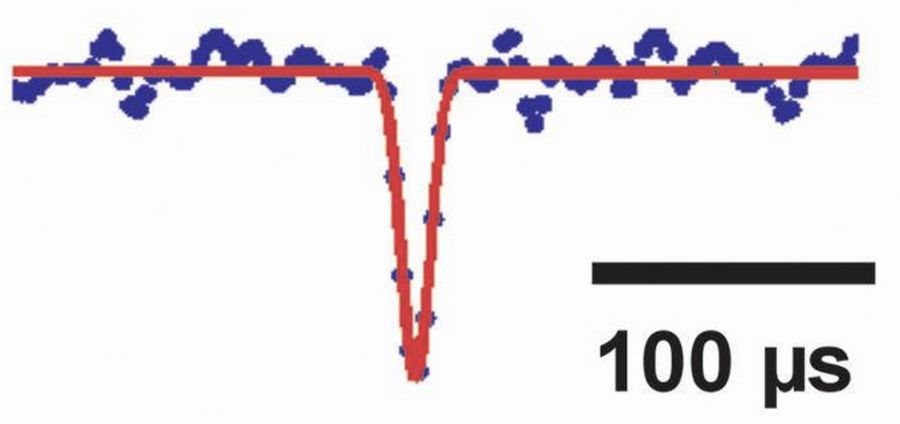
Low-noise single channel recordings with quartz-glass pipettesTo resolve low-noise single channel currents, quartz-glass pipettes are used (Figure showing an opening of a nicotinic acetylcholine channel with a duration of 10 µs; Hallermann et al., J Physiol, 2005). |
 |

